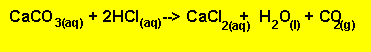A feature of this technique is that we use excess reagent and two stoichiometric equations.We generally use this technique when we are dealing with very weak acids and bases that are unable to make an indicator change colour sharply at the end point of a titration.
Lets see an example, although in this example we are using a strong acid it is just the process that we are concerned about.
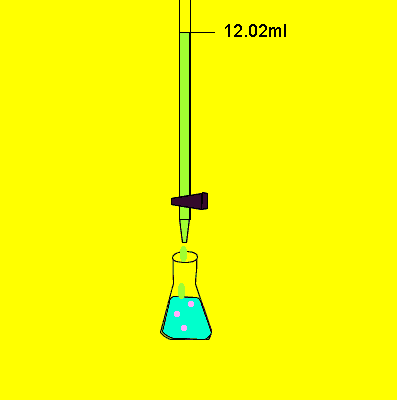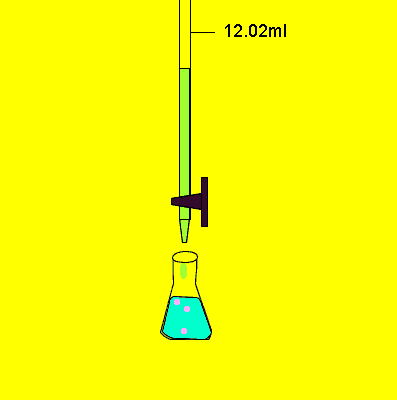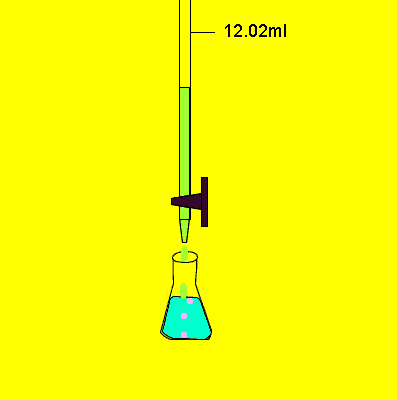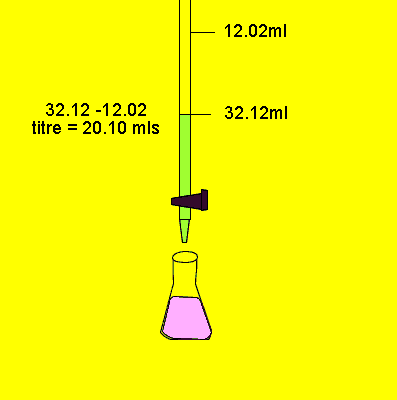
A 20.0g piece of steel wool is dissolved in 300.0mL of 1.00M sulphuric acid, H2SO4. The excess sulphuric acid is determined by titration with a 0.500M NaOH solution, as shown on the right. 280.0mL of sodium hydroxide is required to neutralise the acid. What was the percentage, by mass, of iron in the steel wool?
Step 1) Find the mole of NaOH used to neutralise the excess acid.
(n = c v) 0.500 x 0.280 = 0.140
Step 2 Derive the equation of the reaction between NaOH and H2SO4.
2NaOH(aq) + H2SO4(aq) => Na2SO4(aq) + 2H2O(l)
Step 3 From the equation above find the excess H2SO4.
(n=cv) ½ x 0.140 = 0.0700
Step 4 Calculate the initial amount of H2SO4 .used
(n=cv) 1.00 x 0.300 = 0.300
Step 5 Calculate the amount of H2SO4 that reacted.
0.300 – 0.0700 = 0.230
Step 6 Derive the equation for the reaction between H2SO4 and Fe
Fe(s) + H2SO4.(aq) => FeSO4(aq) + H2(g)
Step 7 Calculate the mol of Fe in steel wool 1/1 x 0.230 =0.230
Step 8 Calculate the mass of Fe (m=n x Fm) 0.230 x 55.8 = 12.83g
Step 9 Calculate the % of Fe (12.83 / 20.0) x 100/1 = 64.2% |