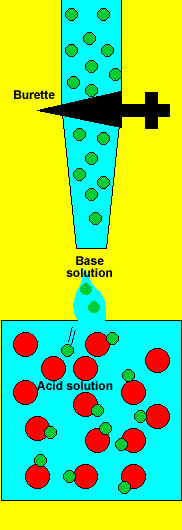
Titration is a chemical procedure that is
used to identify how much substance is dissolved in a given volume. In
other words how concentrated a solution is.
This method is commonly used to test the concentration of acid solutions.
A sample of the acid solution is taken and a chemical that reacts with
the acid(base) is delivered accurately from the burette. Acids react with
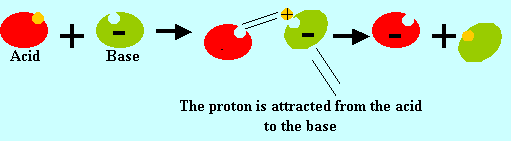
bases to neutralise each other. The reaction is represented below.
The more base that is delivered from the burette the more acid that was present in the volume of test solution.
This procedure is very accurate and care must be taken not to contaminate the equipment with water or other chemicals when cleaning.
"Concentrated solutions of acid are very strong indeed."
Is this correct? Read on and complete this exercise