Robert Boyle (1627 - 1691) investigated the relationship between pressure
and volume of a fixed amount of gas at constant temperature.
His investigations revealed that pressure and volume were inversely
related. This realtionship is known as Boyle's Law.
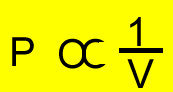
P= pressure
V=volume
We can conduct an experiment in the classroom to investigate this law but for now we will conduct a simple virtual experiment. Using 1kg bricks to apply pressure on the plunger of a glass syringe we will measure the resulting volume and plot the data on a set of axis..
Click to run the experiment.
We can manipulate the expression on the right to form
PV = K
The product of pressure
and volume is a constant for a fixed amount of gas at constant temperature.
So if the pressure and volume of the gas changes the product of pressure
and volume will be the same as shown by the expression below.
P1V1 = P2V2
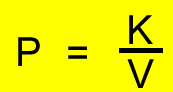
.Let's try some examples. We will use SI units so pressure will be kPa and volume is in litres.
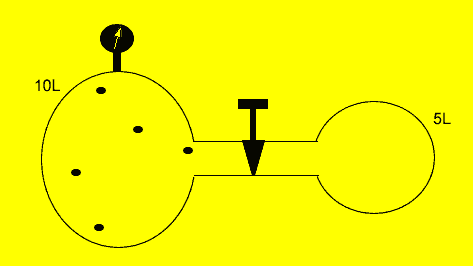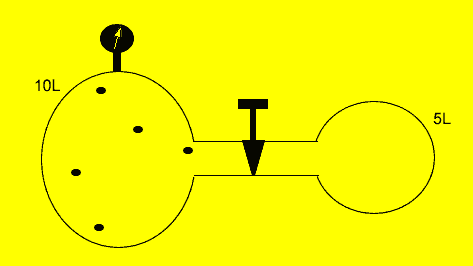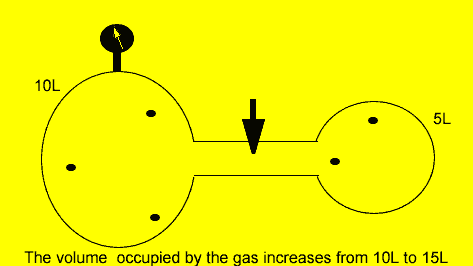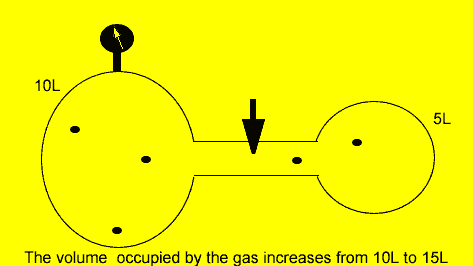
A fixed amount of gas was placed in a 10 litre vessel at a pressure of 101.3kPa. The tap was opened and the gas was allowed to flow freely into another 5 litre chamber as shown on the left.
Calculate the
final pressure inside the vessel if the temperature was kept constant.
Since the amount of gas and the temperature were constant the expression
below is true.
P1V1 = P2V2
P1 = 101.3, V1 = 10L
P2 = ?, V2 = 15L
=> 101.3 X 10 = P2 X 15
=> 101.3 X 10 / 15 =P2
=> 67.53kPa =P2