|
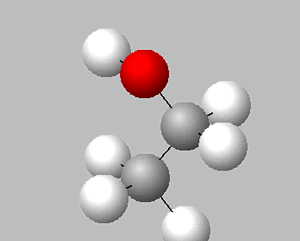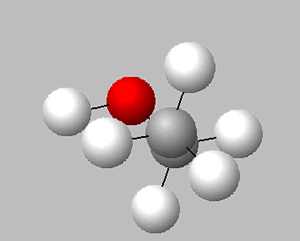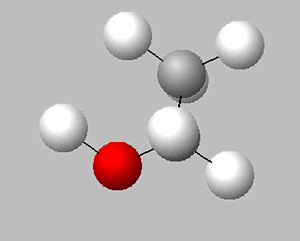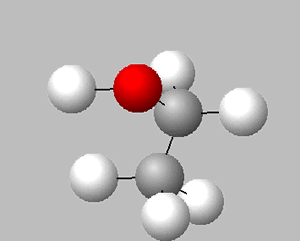
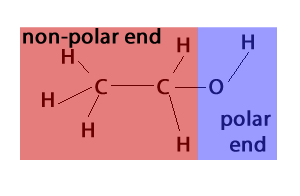
The biological process of fermentation is another source of ethanol. This process is predominantly carried out by microscopic fungi called yeast, pictured on the right. Although the process is complex, we will simplify fermentation by representing it as the overall process by which glucose (C6H12O6) is converted into ethanol (CH3CH2OH) and carbon dioxide gas (CO2).
C6H12O6 => 2CH3CH2OH + 2CO2 + Energy (ATP)
Ethanol is toxic to yeast and can only be tolerated at concentrations below 15% (v/v).
Ethanol is considered a renewable energy source because it is produced by the chemical breakdown of sugars that are produced during the process of photosynthesis. The overall reaction of photosynthesis is shown below.
6H2O + 6CO2 + Energy => C6H12O6 + 6O2
The cellulose in plant matter, such as sugar cane waste, can be broken down by bacteria and the sugars then fermented to produce ethanol. Synthesis of ethanol and subsequent purification, is energy consuming and the thought of ethanol as a viable, green energy alternative is negated by the energy used in its production.
Ethanol as a fuel is mixed with petrol where 85% of the fuel is composed of ethanol. By 2009 over six million “dual-fuel” ethanol-compliant cars and trucks, had been manufactured in America, however, a lack of filling stations means that most of these vehicles run on petrol most of the time.
The combustion of ethanol, shown below, releases heat energy which drives the pistons of the vehicle.
- CH3CH2OH + 3 O2 → 2 CO2 + 3 H2O + heat

Image from Google
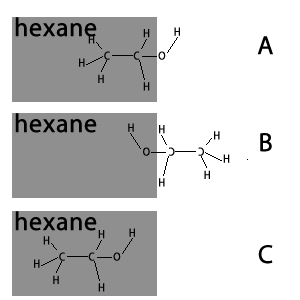
Hexane is a non-polar substance. Which of the following is the orientation of ethanol in hexane?
Explain.

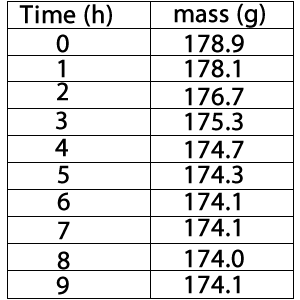
The table on the right shows the data the student recorded.
a) What are the dependent and independent variables in this experiment?
b) Graph the results on an appropriate set of axis.
i) What will represent the y- axis?
ii) What will represent the x-axis?
c) What do you attribute to the mass loss?
d) How many hours into the experiment does the rate of fermentation peak?
e) Why does mass loss plateau after 6 hours of fermentation?
f) What volume of gas was produced by the yeast at SLC during the 9 hours of fermentation?