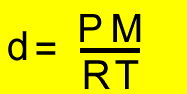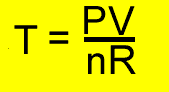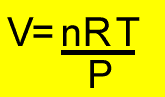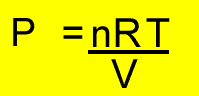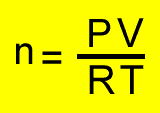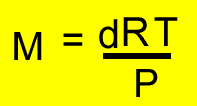Manipulating
the ideal gas equation
PV =nRT
PV =nRT

n = mole
d = density (g/L)
M = molar mass of the gas under investigation
P = pressure (kPa)
T = temperature(K)
V = volume(litres)
R = 8.31J/K/mol
The best way to tackle the ideal gas equation is to get straight into using it. Click on a formula in the column on the right to try several exercises using that particular formula.