Starch and cellulose
Starch is a polymer used to store glucose while cellulose is a polymer of great structural importance. Both giant molecules are formed by a condensation polymerisation reaction with the loss of water.

|
Starch and cellulose |
|
Starch is a polymer used to store glucose while cellulose is a polymer of great structural importance. Both giant molecules are formed by a condensation polymerisation reaction with the loss of water. |
|
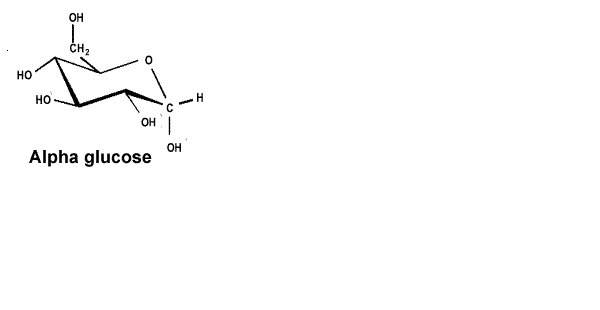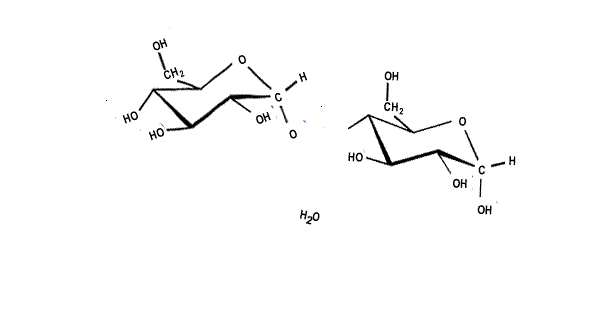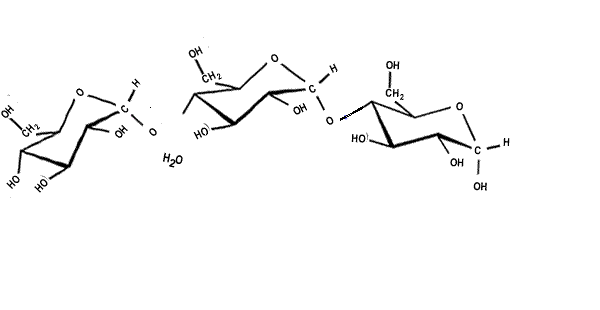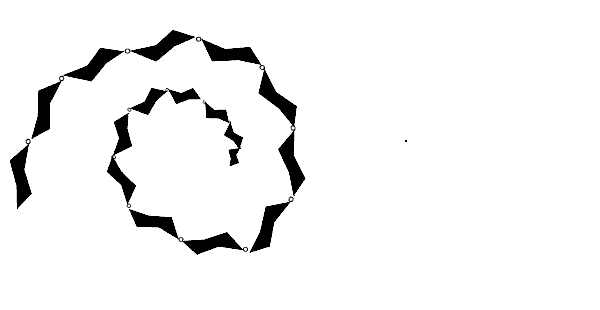
| When two sugar molecules, monosacharides (C6H12O6) bond a single molecule of water is removed forming a covalent bond which results in a glycosidic link. Glycosidic links are also know as ether links. This forms a disaccharide with a general formula C12H22O11, as shown on the right. |  |
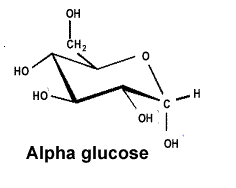
| Starch is a polymer formed from alpha glucose, shown on the right. |  |
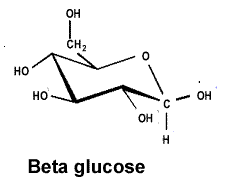
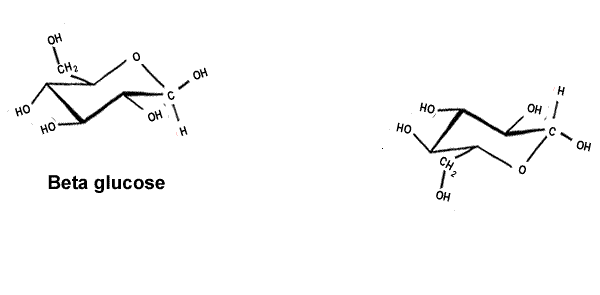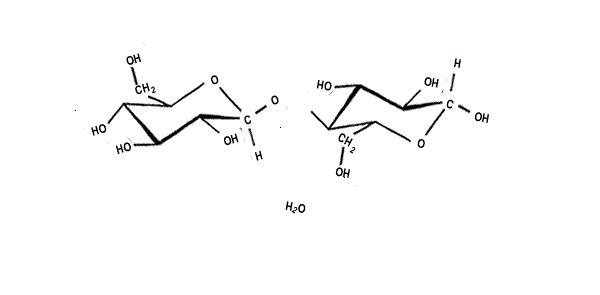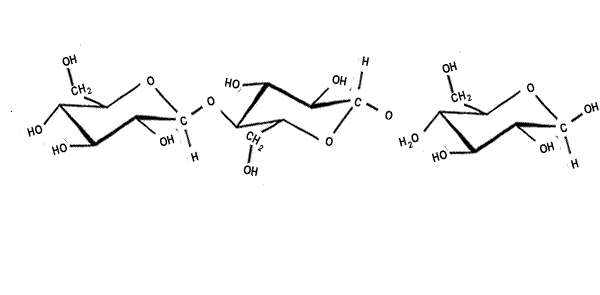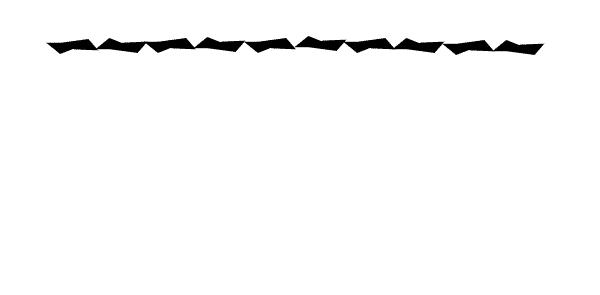
| Cellulose is a polymer formed from beta glucose, shown on the right. |  |
| The alpha and beta glucose molecules are known as isomers. Isomers are molecules that have the same molecular formula but a different structural formula. Although the difference between the two isomers is subtle the difference in the polymers formed is stunning. | |
| Starch is a coiled polymer that is used by plants to store glucose. The animation below shows the condensation polymerisation reaction to form a small section of a starch polymer. | |
 |
|
| Cellulose is a linear polymer, of around 10,000 glucose units long, that packs tightly with other cellulose polymers. Below is an animation of the polymerisation of beta glucose to form a small section of a cellulose polymer. Notice how every second beta glucose molecule is flipped upside down. | |
 |
|