The explosive chemistry
of nitrogen, phosphorus and urine.
Nitrogen is an essential component of living matter. It is present in protein molecules that form all organisms. Unlike oxygen, nitrogen can not be used by animals and plants directly from the air. Even though 78% of the atmosphere is due to nitrogen gas its availability is extremely limited. Plants must absorb nitrogen in the form of ammonium ions (NH4+) and nitrate ions (NO3-) in order to build the complex molecules that we know as amino acids, the building blocks for proteins. Conversion of nitrogen gas into these ions is difficult and can occur only at high temperatures or by bacteria in the soil. This process is called nitrogen fixing.
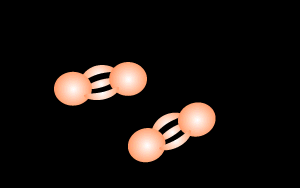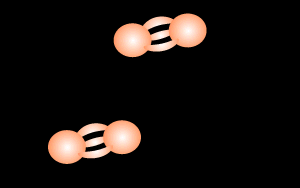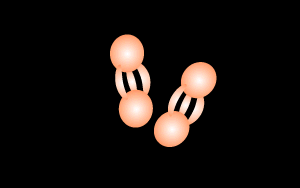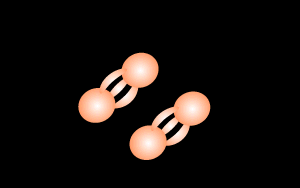
During lightning strikes enough energy is supplied to break the bonds of the nitrogen molecule and form nitrous oxide according to the equation below.
N2 + O2
=> 2NO
The nitrous oxide formed combines with oxygen to form nitrogen dioxide
according to the equation below.
2NO + O2 => 2NO2
Nitrogen dioxide readily dissolves in water to produce nitric and nitrous
acids which provides a source of nitrates available to plants.
2 NO2 + H2O => HNO3 + HNO2
Nitrogen fixation is also carried out by bacteria in the soil according to the reaction below.
N2 + 8H+ => 2 NH4+ (ammonium ion)


Nitrates, such as potassium nitrate (saltpeter) and sodium nitrate are mixed with fuels to form very explosive mixtures. Evidence of the immense explosive power of mixtures of nitrates and fuel are the ruins of the Alfred P. Murrah Federal Building in Oklahoma City. As seen on the right, the building was destroyed by a truck loaded with fertiliser (ammonium nitrate) and fuel (petrol).

The use of saltpeter was first documented in ancient Greece where Greek-fire was reported to have originated. Greek-fire was a mixture of saltpeter, sulfur, charcoal and other flammable substances. Once set on fire the mixture burnt fiercely, even under water.

In 1245 Friar Roger Bacon mixed
saltpeter, carbon and sulfur and made gunpowder. Gunpowder involves a
very powerful exothermic reaction as outlined below.
16 KNO3(s)
+ S8(s) + 24 C(s) => 8 K2S(s)
+ 24 CO2(g) + 8 N2(g)
The supply of saltpeter was limited and the munitions industry was unable
to get enough to meet demand.
Saltpeter is now mined in huge quantities. Before WW1, Europe depended on imports of nitrates (saltpeter) for weapons production. Without its own source of nitrates, Germany found itself in a very weak position. British domination of the seas prohibited Germany to receive imports of nitrates in quantities needed for munitions manufacture. A German chemist, Haber, changed the situation and made Germany self-sufficient in nitrogen compounds through a process of making ammonia from hydrogen and nitrogen gases. This is called the Haber process and the critical reaction is given by the equation below.
N2(g) + 3H2(g) <=> 2NH3(g)
From ammonia, nitric acid is formed according to the equation below in a process called the Ostwald process.
NH3(g) + 2O2(g) => HNO3(aq) + H2O(l)
After the synthesis of nitric acid formation of sodium nitrate, ammonium nitrate and saltpeter was easily accomplished according to the equations below. The Haber process and the Ostwald process revolutionised the explosives industry.
HNO3 + NH3 => NH4NO3
HNO3 + NaOH =>NaNO3 + H2O
HNO3 + KOH => KNO3 + H2O
78% of the atmosphere is due to nitrogen gas. Considering this fact, explain why the soil is relatively poor in nitrogen compounds?
Explain how Haber and Ostwald may have contributed to Germany's rise as a military power.
Describe the composition of the mixture known as Greek fire. Why was it difficult to extinguish the burning mixture?
Senior chemistry question
Hide
Consider the reaction of gunpowder below.
What is being reduced?
What is being oxidised?
Name the reductant
Name the oxidant
16 KNO3(s) + S8(s) + 24 C(s) => 8 K2S(s) + 24 CO2(g) + 8 N2(g)
Solution
Hide
Sulfur (S8 ) is reduced from an oxidation state of 0
to -2 in K2S
Carbon is oxidised from an oxidation state of 0 to +4 in CO2
The reductant is carbon
The oxidant is sulfur