1. Which one of the following shows the 13C NMR spectrum of
pentan-3-one, C5H10O?
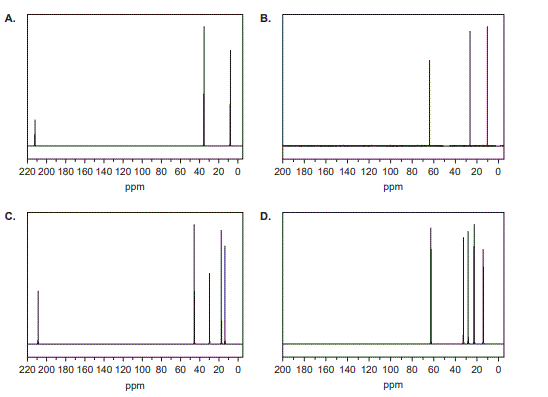
Solution

2. A sample separated from a mixture of compounds produced the IR spectrum shown below.
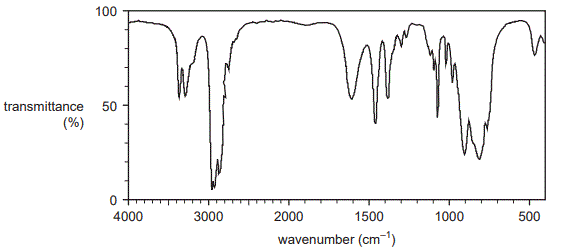
State whether this spectrum was produced by an amine or a carboxylic acid.
(
1 mark)
Solution

3. Compound X, known to contain both oxygen and chlorine, was purified from a waste sample to enable it to be re-used.
a. The mass spectrum of Compound X is shown below:
• The only stable isotope of oxygen is 16O.
• The two naturally occurring, stable isotopes of chlorine are 35Cl and 37Cl.
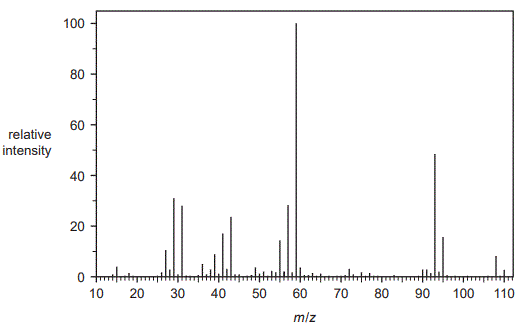
i. Identify the m/z of the base peak. (1 mark)
ii. State why all the possible isomers of Compound X would have the same peak at m/z = 108. (1 mark)
iii. Identify the formula of the species responsible for the peak at m/z = 95.
(1 mark)
Solution.

b. The high-resolution 1H NMR spectrum for Compound X is shown below.
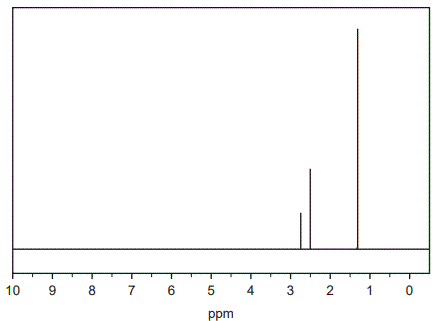
Data obtained from the 1H NMR spectrum for Compound X is shown in the table below.
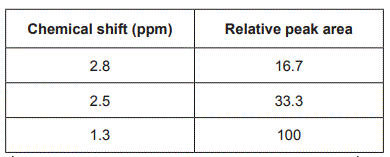
Referring to the 1H NMR spectrum and the table above, deduce three things about the structure of Compound X. (3 marks)
Solution

c. The 13C NMR spectrum for Compound X is shown below.
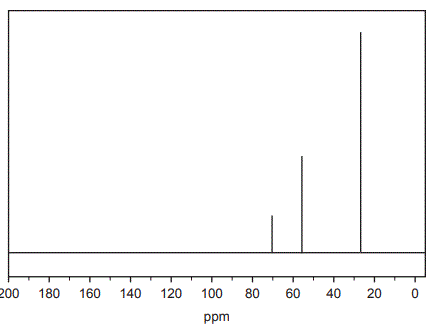
What does the 13C NMR spectrum indicate about the structure of Compound X? (2 marks)
Solution
d. i. In the space provided below, draw the skeletal diagram of Compound X that is consistent with the information provided in parts c–e. (1 mark)
ii. Write the IUPAC name of Compound X. (1 mark)
Solution
