1. The reaction to produce methanal, CH2O, is shown below.

The primary role of the catalyst in the production of CH2O is to increase the
A. speed of all particles.
B. number of collisions per unit time.
C. proportion of particles that react.
D. overall kinetic energy of the system.
Solution
2. The major organic product formed when propan-1-ol,
CH3CH2CH2OH, reacts with butanoic acid,
CH3CH2CH2COOH, in the presence of sulfuric acid, H2SO4, is
A. CH3CH2COOCH2CH2CH2CH3
B. CH3CH2CH2CH2OCOCH2CH3
C. CH3CH2OCOCH2CH2CH3
D. CH3CH2CH2OOCCH2CH2CH3
Solution

3. In a protein hydrolytic reaction, which one of the following combinations is correct?
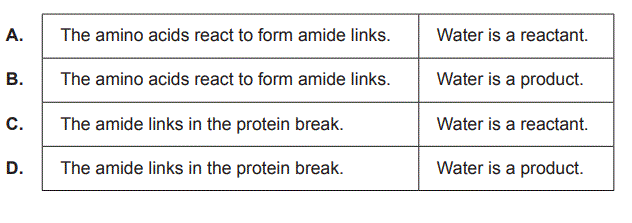
Solution
4. During fermentation, yeast will produce other volatile polar compounds that have similar boiling points to ethanol, C2H6O. Which one of the following methods would be most suitable to separate these compounds from C2H6O?
A. solvent extraction
B. simple distillation
C. fractional distillation
D. solvent extraction and distillation
Solution
5. Geranial and linalool are two natural organic compounds found in essential oils extracted from some citrus fruits. The skeletal structures for geranial and linalool are given below.
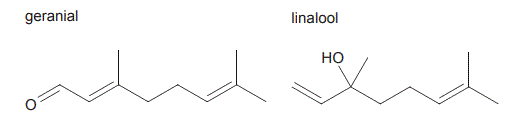
a. The molecular formulas for geranial and linalool are
A. C10H15O and C10H17O
B. C10H15O and C10H18O
C.C10H16O and C10H18O
D. C10H16O and C10H17O
Solution
b. Which one of the following laboratory tests could be used to confirm that a sample is pure geranial and not pure linalool?
A. React the sample with acidified dichromate ion, H+ /Cr2O72- . The colour of the solution will change from orange to green.
B. React the sample with acidified permanganate ion, H+ /MnO4- . The colour of the solution will change from pale pink to purple.
C. React the sample with bromine solution. The colour of the solution will change from brown to colourless.
D. React the sample with solid sodium hydrogen carbonate, NaHCO3. Bubbles will be produced.
Solution
6. Vanillin is a widely used ingredient in the food industry. Pure vanillin has a melting point of 82–83 °C. To reduce costs, it is often blended with significant amounts of cheaper compounds that have similar melting points.
A blended sample was tested. The melting point range of the blend would be closest to
A. 68–79 °C
B. 79–80 °C
C. 82–83 °C
D. 100–109 °C
Solution
7. One mole of a triglyceride containing only one type of fatty acid reacts completely with 6 moles of iodine, I2. Which one of the following could be the fatty acid?
A. linolenic acid
B. linoleic acid
C. oleic acid
D. stearic acid
Solution


8. Which one of the following shows the 13C NMR spectrum of
pentan-3-one, C5H10O?
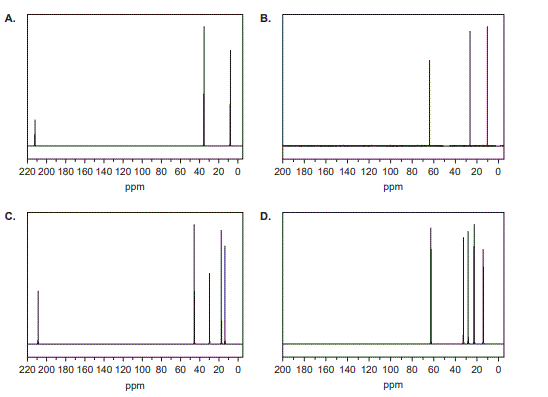
Solution

9. Many industries, such as the pharmaceutical industry, need to produce a range of organic compounds.
a. Simple hydrocarbons are the usual raw material used to produce haloalkanes, amines, alcohols, carboxylic acids and esters.
i. Write a balanced equation to show the production of C3H7Cl from C3H8. Include the reaction conditions.(2 marks)
Solution
ii. Name two reactants required to form propan-1-amine, C3H9N, in a single substitution reaction. (2 marks)
Solution
During the production process, routine quality testing occurs. After testing, the samples are collected as organic waste. In order to dispose of the waste safely, the components must be separated.
b. The waste from one laboratory was a mixture of amines and carboxylic acids. The amines were separated from the carboxylic acids on the basis of their different boiling points. Boiling points of the first three compounds in the carboxylic acid and amine series are listed in the table below.
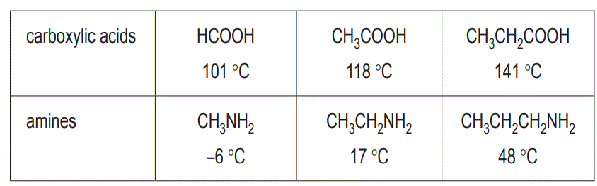
i. Referring to their structures, explain why the homologous series of carboxylic acids have higher boiling points than the corresponding amines. (2 marks)
Solution
One of the samples that was separated from this waste mixture produced the IR spectrum shown below.
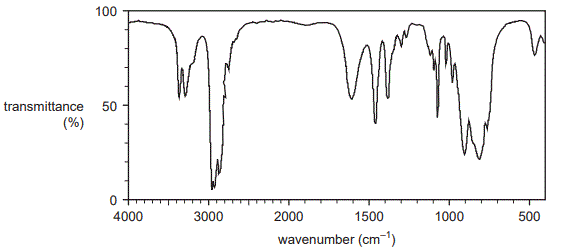
ii. State whether this spectrum was produced by an amine or a carboxylic acid.
(
1 mark)
Solution

Compound X, known to contain both oxygen and chlorine, was purified from a waste sample to enable it to be re-used.
c. The mass spectrum of Compound X is shown below:
• The only stable isotope of oxygen is 16O.
• The two naturally occurring, stable isotopes of chlorine are 35Cl and 37Cl.
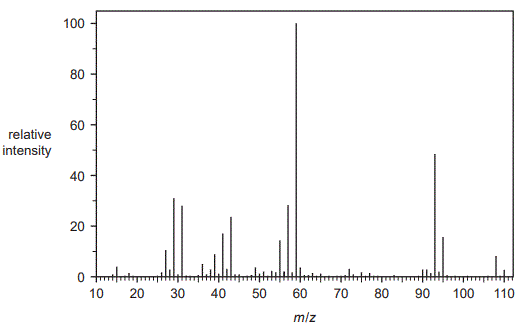
i. Identify the m/z of the base peak. (1 mark)
ii. State why all the possible isomers of Compound X would have the same peak at m/z = 108. (1 mark)
iii. Identify the formula of the species responsible for the peak at m/z = 95.
(1 mark)
Solution.

d. The high-resolution 1H NMR spectrum for Compound X is shown below.
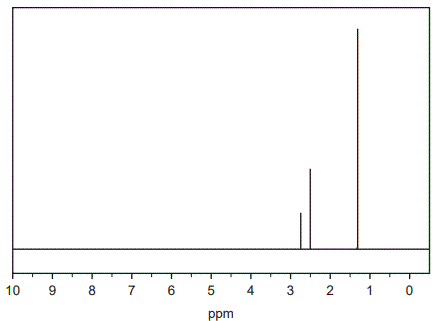
Data obtained from the 1H NMR spectrum for Compound X is shown in the table below.
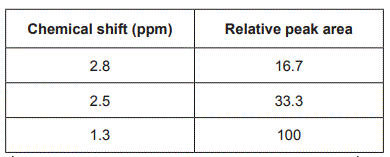
Referring to the 1H NMR spectrum and the table above, deduce three things about the structure of Compound X. (3 marks)
Solution

e. The 13C NMR spectrum for Compound X is shown below.
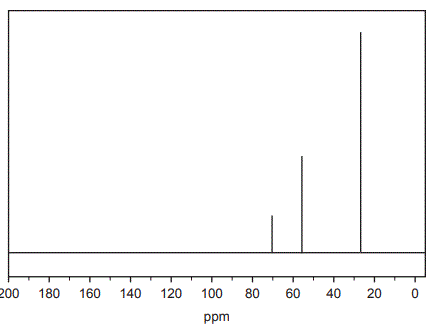
What does the 13C NMR spectrum indicate about the structure of Compound X? (2 marks)
Solution
f. i. In the space provided below, draw the skeletal diagram of Compound X that is consistent with the information provided in parts c–e. (1 mark)
ii. Write the IUPAC name of Compound X. (1 mark)
Solution

10. A representation for quinine is shown below with three functional groups circled.
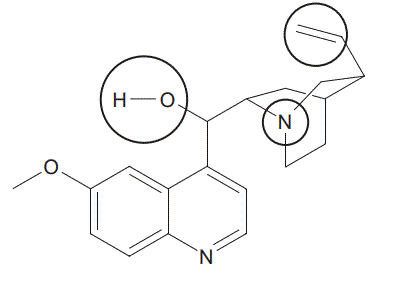
a. Using item 19 of the Data Book, name the functional group with the highest priority when following IUPAC nomenclature. (1 mark)
Solution
b. Malaria is caused by the malaria parasite, which enters red blood cells and feeds on haemoglobin. Experiments have shown that the malaria parasite enzymes involved in this reaction are most effective at a pH of 4.7 The structure of the quinine molecule can vary with pH. Representations of two possible structures are shown below.
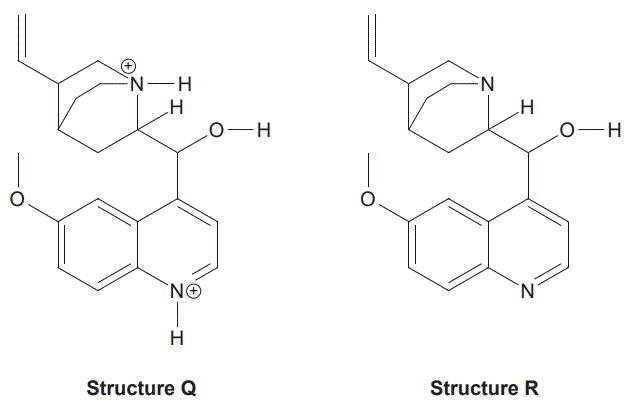
Identify which structure, Q or R, is most likely to occur at a low pH. Justify your response. (2 marks)
Solution

c. Quinine has four chiral centres. Only one of its isomers is an effective medicine in the treatment of malaria.
i. Referring to the term ‘chiral centre’ in your response, explain why only one isomer of quinine is effective. (3 marks)
Solution
ii. Quinine acts as a competitive inhibitor in the prevention of malaria. Explain why quinine is more effective as a competitive inhibitor at pH 4.7 than at a higher pH. You may draw a diagram to support your response. (2 marks)
Solution
