1) Which one of the following correctly identifies the renewability of biomethane and natural gas?
Solution
2)
A 65.0 g snack bar contains:
• 45.0 g of carbohydrate
• 10.0 g of protein
• 10.0 g of fats.
The total energy content per 100 g of the snack bar is closest to
A. 1.3 × 103 J
B. 1.9 × 103 J
C. 1.3 × 106 J
D. 1.9 × 106 J
Solution
3) The circular economy of a bioethanol production process could be improved by
A. increasing the fermentation temperature to accelerate the reaction rate.
B. using a new strain of yeast to produce a higher yield of ethanol from glucose.
C. developing a separate process that converts waste carbon dioxide into a useful product.
D. carrying out advanced distillation techniques to reduce energy consumption.
Solution

4)The production of bioethanol from sugar cane follows the steps shown below. 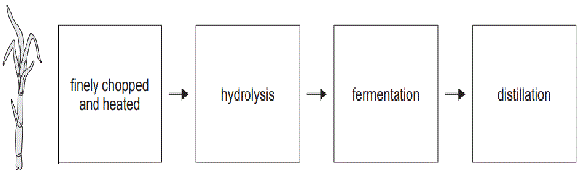
Consider the following statements:
I Oxidation occurs during fermentation.
II Enzymes catalyse only two of the steps shown.
III Glucose is a product of the hydrolysis step.
Which of the statements about the production of bioethanol is/are correct?
A. II only
B. I and II
C. I and III
D. II and III
Solution

5) Kim investigated the energy efficiency of two different fuels, propane, C3H8, and pure bioethanol, C2H5OH, for heating water. Kim placed a saucepan with 166.0 g of tap water on a portable camp stove and heated it using C3H8. The set-up for the experiment is shown below.
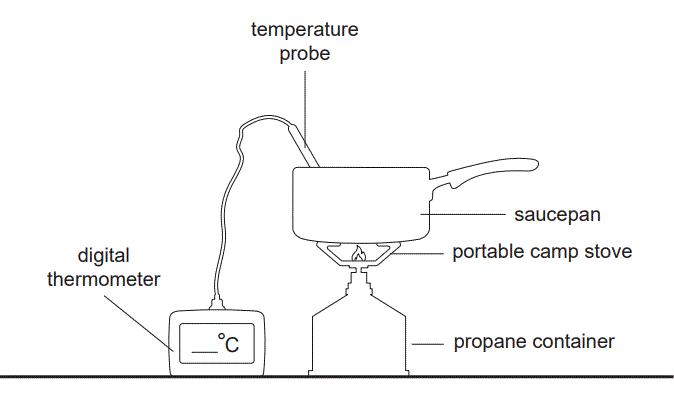
The following results were recorded.

a. Write a balanced thermochemical equation for the complete combustion of
C3H8. (2 marks)
Solution
b. Using item 13 of the Data Book, calculate the amount of energy released by the complete combustion of 9.0 g of C3H8 (M = 44.0 g mol−1 ). (2 marks)
Solution

c. i. Calculate the energy absorbed by the water in the saucepan. (2 marks )
ii. Determine the overall energy efficiency of heating the water in the saucepan.
(1 mark)
Solution

d. Kim placed all of the equipment inside a sealed container with a total air volume of 0.125 m3 and repeated the investigation. The air in the enclosure contained 21.0% v/v oxygen, O2, at standard laboratory conditions (SLC).
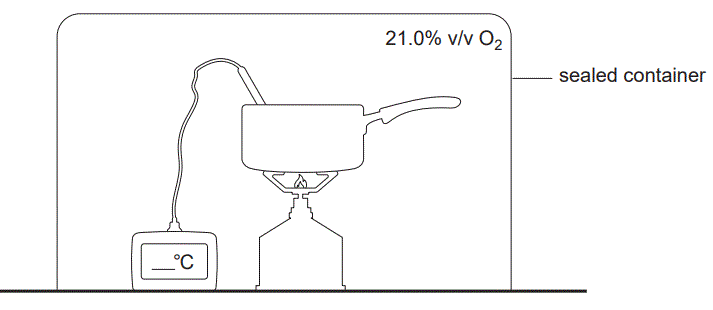
9.0 g of C3H8 was combusted in the sealed container shown above. Determine the mass of the reactant in excess. (4 marks)
Solution

e. In another investigation, Kim found that 15.3 g of C2H5OH (M = 46.0 g mol−1 ) was required to produce the same change in water temperature as the 9.0 g of C3H8. Explain why different masses of the fuels were required to produce the same temperature change. (2 marks )
Solution