1. Consider a fuel cell using gaseous reactants. Which one of the following design features would significantly enhance fuel cell efficiency?
A. including a dense, non-porous electrode to limit gas diffusion
B. using liquid electrolytes to transport reactants between the electrodes
C. incorporating porous electrodes to maximise the surface area for catalytic reactions
D. using a solid, impermeable membrane between electrodes to minimise gas loss between half-cells
Solution
2. A fuel cell utilises the reaction between hydrogen, H2, and oxygen, O2, to produce water, as shown in the reaction below.
2H2(g) + O2(g) → 2H2O(l)
When a fuel cell produces 36000 C of charge, the mass of O2 consumed is closest to
A. 11.9 g
B. 2.98 g
C. 1.49 g
D. 0.373 g
Solution
3. In innovative fuel cells, like the electrolyte-free (layer-free) fuel cell (EFFC), the internal cathode–electrolyte–anode structure commonly found in conventional fuel cells is replaced with a single conductive porous layer, as shown in the diagram below.
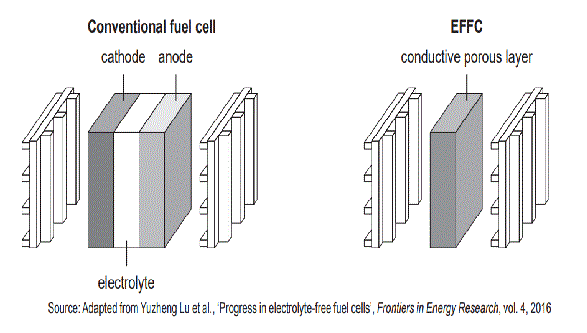
Manufacturers of the EFFC claim the following benefits:
I. The single porous layer can be made from material with superior conductive properties.
II. Fewer internal interfaces, compared to conventional fuel cells, result in less heat loss.
Which of the claims above describes how the operation of the EFFC aligns with the green chemistry principle of design for energy efficiency?
A. I only
B. II only
C. both I and II
D. neither I nor II
Solution
