1. due to the range of different compounds contained in its bark. One of the useful medicinal compounds in its bark is quinine, C20H24N2O2, which is used to make tonic water. Tonic water sold today has a much lower concentration of quinine than in the past. a. High-performance liquid chromatography (HPLC) is commonly used to detect both the presence and concentration of quinine in tonic water. The HPLC chromatograph of a sample of tonic water is shown below.
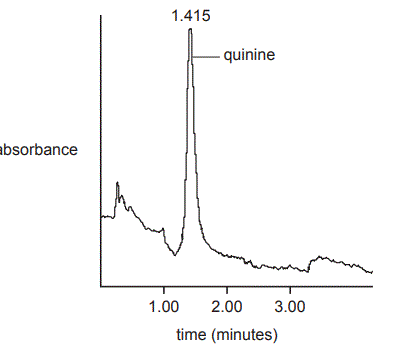
i. Explain how retention time can be used to confirm the presence of quinine in the sample of tonic water. (2 marks)
Solution

Various concentrations of quinine solutions were run through an HPLC instrument to produce the calibration curve shown above. A sample of tonic water is to be tested to determine the concentration of quinine.
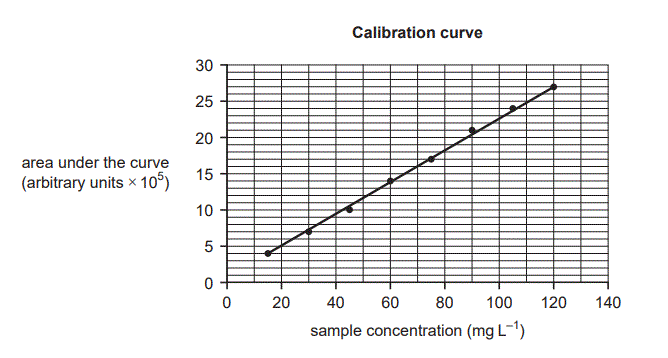
ii. Explain why solutions of known concentration must be run through the HPLC instrument under controlled conditions. (2 marks)
Solution

iii. State why the calibration curve should not be used to test a sample of tonic water that has a mean concentration of 130 mg L−1 . (1 mark)
Solution
