1) The table below gives the heat content, in kilojoules per gram, of a number of different fuels.
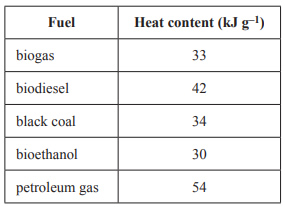
Given the information above, it can be stated that
A. petroleum gas is pure methane.
B. all of the fossil fuels produce more energy by mass than the renewable energy sources.
C. the renewable energy source that produces the most energy by mass is biodiesel.
D. biogas can be produced from renewable energy sources and produces more energy by mass than petroleum gas.
Solution

2) Different types of fuels can be used in vehicles with combustion engines. These fuels can be produced from either fossil fuels or from renewable sources. Liquefi ed petroleum gas (LPG), a fossil fuel used in cars, is mainly made up of propane,
C3H8.
a) Write a balanced thermochemical equation for the complete combustion of C3H8 in air at standard laboratory conditions (SLC).
Solution

b) How much energy, in kilojoules, would be produced from the complete combustion of 290 g of C3H8 at SLC?
Solution.

c) What volume of air (21.0% oxygen, O2, by volume), measured at SLC, would be required to fully combust 68.5 g of C3H8?
Solution

3) Biodiesel is a renewable fuel produced from a reaction between an alcohol and a triglyceride. The nature of both the fatty acids in the triglyceride and the alcohol involved affects the viscosity of the fuel produced. A scientist produces a variety of alkyl esters from different combinations of fatty acids and alcohols that can be used as fuels. The viscosity of these fuels is measured at 40 °C, in arbitrary units, and listed in Table 1 below. When measured in these arbitrary units, the larger the value the greater the viscosity.
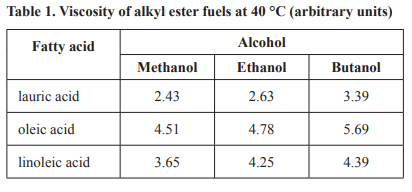
d) According to the data provided, which combination of alcohol and lauric acid produces a biodiesel that fl ows the slowest through a fuel line at 40 °C?
e) Oleic acid and linoleic acid contain the same number of carbon atoms. The viscosities of the esters of linoleic acid are consistently lower than the viscosities of the corresponding esters of oleic acid, as shown in Table 1. What feature of linoleic acid contributes to its lower viscosity? Justify your answer
Solution
