
View the video on the right. See how water boils as the air pressure inside the syringe is lowered.
We can simply state what is happening in the video by saying that more pressure on the surface of the water means that the water vapour has to push harder on the air above the surface of the water and less pressure means that there is less air to push out of the way and so it is easy for bubble of water vapour to escape.
When it is harder for water vapour to push to the surface due to greater pressure a lot more heat is needed. Heat gives the vapour particles extra energy with which to move about with greater speed and push to the surface.
Because when we increase our altitude we generally find an accompanying decrease in atmospheric pressure, changing altitude can change a boiling point.
 |
Another
demonstration can be conducted by boiling water in a flask. Allow
the water to boil for a little while so that most of the air is
driven out of the flask. Stopper the flask and turn it upside
down on the bench. Place an ice cube on its base and watch the
water inside the flask boil. The
ice cube causes the air pressure inside the flask to drop. |
| High on top of Mt Everest it is impossible to make a hot cup of tea. Explain Why | |
| After conducting the above demonstration, what two factors influence the boiling point of water? | |
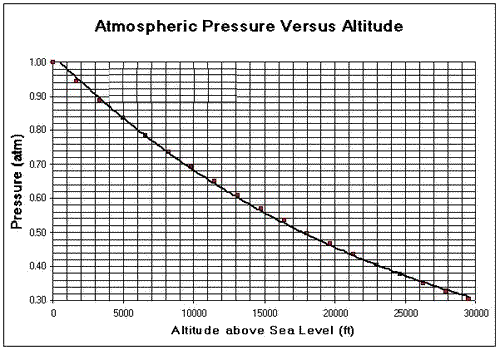 |
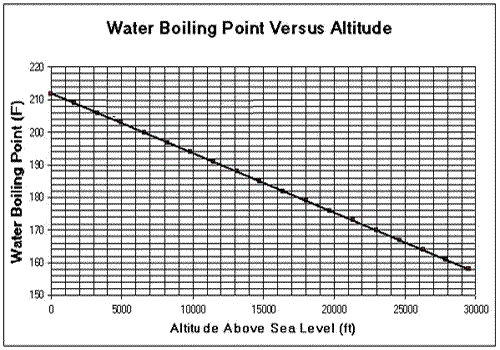
Consider the two graphs shown on the left. The units are in Fahrenheit and feet. Conversions between units must be done. a) The water in the pipe of an aeroplane starts to boil at a temperature of 170o F. i. What is the altitude at which the plane is flying in metres? ii. What is the air pressure outside the plane in atm? b) At what temperature, in Celsius, will the water start to boil if the atmospheric pressure drops to 0.4 atm? c) A scientist is climbing a mountain. He is asked to measure the air pressure at a certain recording station high above sea level. Without the right equipment the scientist has no way of calculating the air pressure at the particular altitude. He does, however, have a kettle, water and a thermometer. He heats the water in the kettle which starts to boil at 80 oC i. What is his altitude above sea level in feet?. ii. What is the external air pressure in atm? |
 |
|