This topic was adapted from an article written in NewScientist 6 February 2010, by Edwin Cartlidge titled "A drop of the weird stuff"
Water has some peculiar and weird properties that are hard to explain with our current understanding of the behaviour of water molecules in the liquid state.
A chance discovery was made by Pettersson and Nilsson, in 2000, while using a very high powered X-ray source to investigate the amino acid glycine. They soon found that the spectrum produced by the water in the glycine solution was far more interesting than the amino acid.
Further X-ray investigation revealed the existence of two separate structures, an ordered, tetrahedral structure and a disordered structure.
Before we continue it must be stated that this new theory is not without its critics.

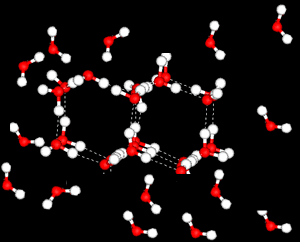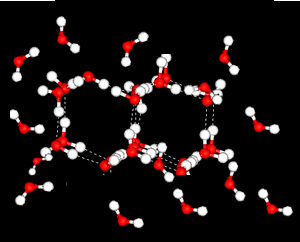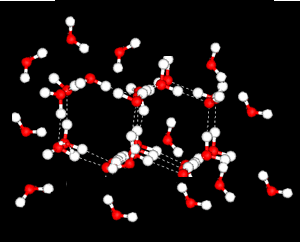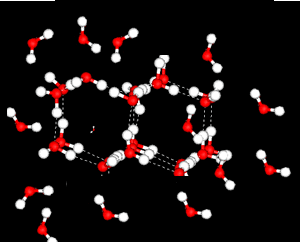
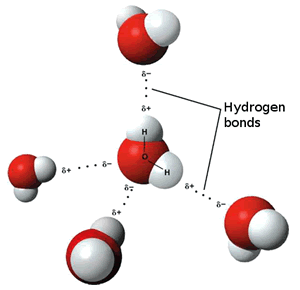
Water is most dense at 4oC.
This is because
heating reduces the number of the ordered, tetrahedral structures in favour of a greater disorder where molecules are more closely packed. However as temperature increases the molecules are forced further apart and density decreases.
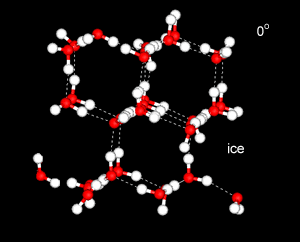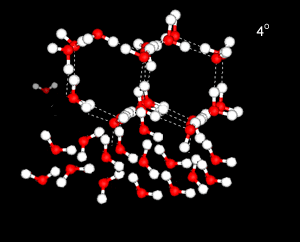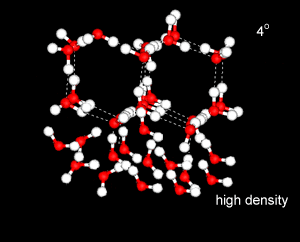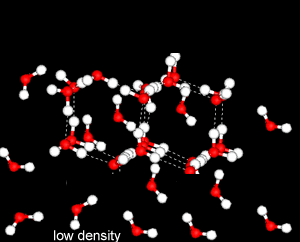
Water has a strangely high specific heat capacity. That is, it takes a great deal of energy to raise the temperature of the liquid by a given amount.
This is due to much of the heat energy going into breaking down the ordered, tetrahedral structures, rather than into increasing the kinetic energy of the water molecules.
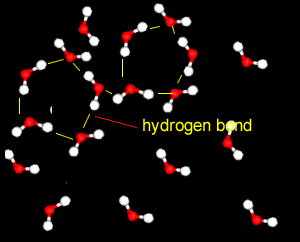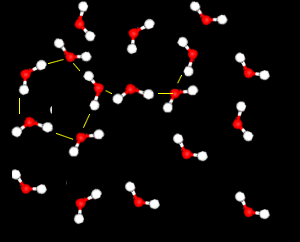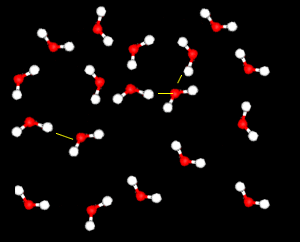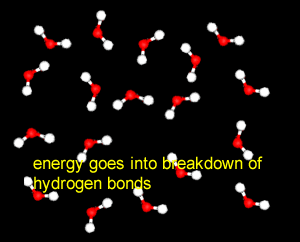
Water is particularly difficult to compress.
This is because the strong attraction between water molecules due to hydrogen bonding, keeps them closely packed unlike the molecules of most other liquids. This effect is particularly noticeable when the disordered structure dominates.
Water's ability to be compressed decreases as the temperature increases until it reaches a minimum at around 45oC. Unlike most other liquids whose compressibility increases with increase in temperature.
This is due to the breakdown of the ordered, tetrahedral structures as the temperature increases which gives rise to more of the disordered regions which are harder to compress. However, at temperatures above 45oC the molecules in the disordered regions are forced apart to such an extent that compressibility increases with increase in temperature.
Unlike most other liquids water becomes less viscous with increase in pressure.
At high pressure the breakdown of the ordered, tetrahedral structures is favored. Molecules are free to move about in the disordered structures than they are in the ordered, tetrahedral structures