Potential difference.
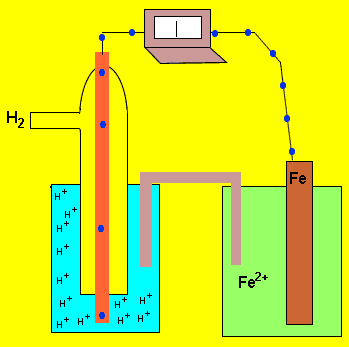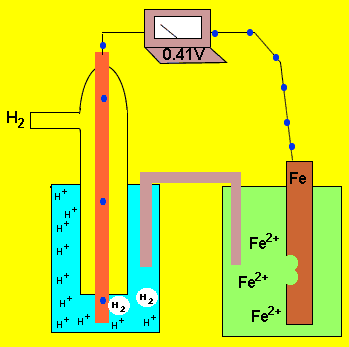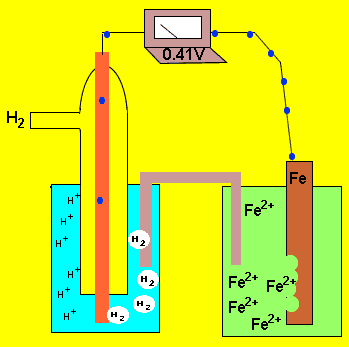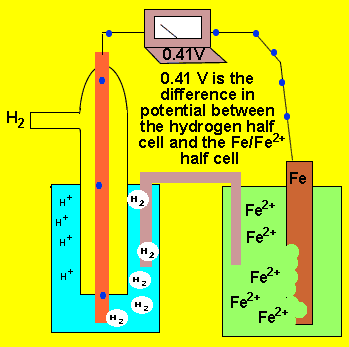
An electrochemical cell is formed by two half cells. A current flows from one half cell to the next because one half cell has a greater capacity to push electrons into the external circuit than the other. The difference in the ability of each cell to push electrons into the external circuit is called the potential difference. This potential difference is sometimes called the emf. This potential difference is measured in volts using a voltmeter and is the measure of energy supplied by a fixed amount of charge flowing through the circuit..
The animation on the left shows a potential difference of 0.41V between
the ![]() half cell and the
half cell and the
![]() half cell.
half cell.