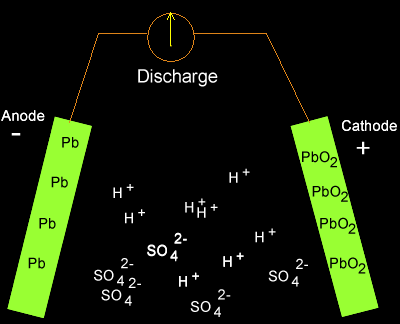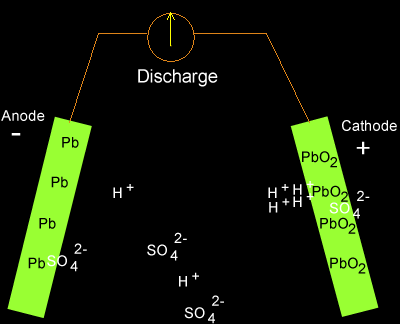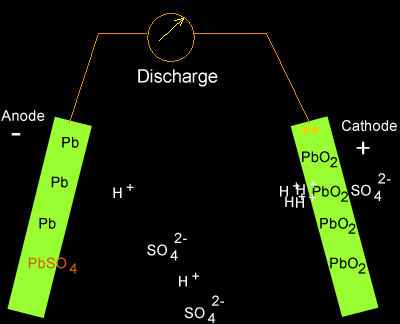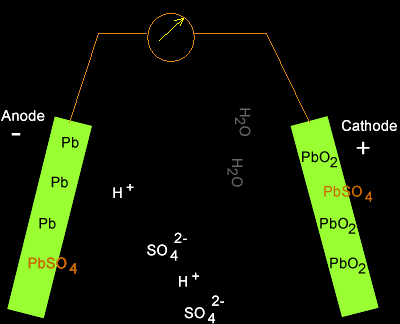
Solution
The overall reaction
when the cell is discharging is
PbO2(s) + 2SO4-2(aq) + 4H+(aq) + Pb(s) => 2PbSO4(s) + 2H2O(l)
As the cell discharges
hydrogen ions are being removed so the concentration of hydrogen ions in
the electrolyte is decreasing. The pH is therefore increasing.
keep in mind that a solution with a pH of 12 has significantly less hydrogen
ions in a given volume than a solution of pH 2.
Translating the pH into figures we can see what really happens.
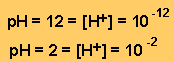
Recharging of the battery produces the following reactions
PbSO4(s)
+ 2H2O(l) => PbO2(s) + SO4-2(aq)
+ 4H+(aq) + 2e
.
This is a oxidation reaction as electrons are given off and will occur at
the anode.
While the reaction below is a reduction reaction (electrons used) and will
occur at the cathode.
PbSO4(s)
+ 2e => Pb(s) + SO4-2(aq)
Click to hide the solution