Catalysts and activation energy
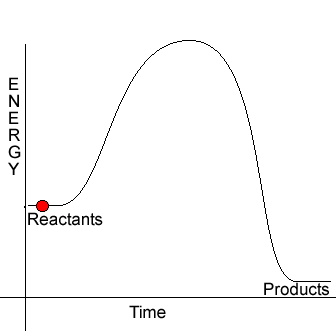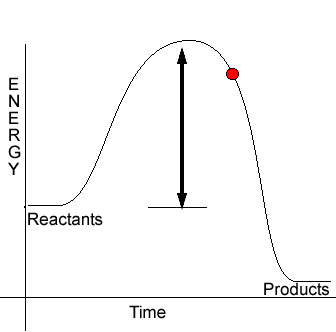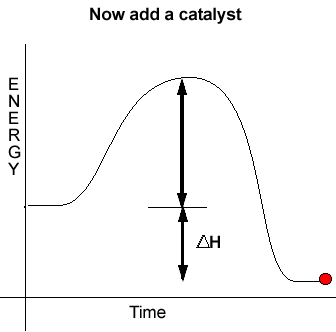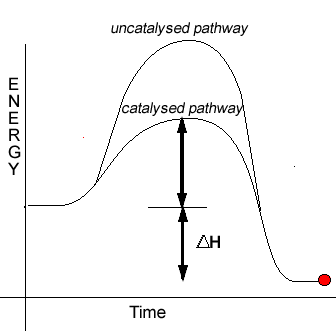
In order for reactions to proceed we must provide energy to break bonds. This energy is known as the activation energy for a particular reaction. It can be represented in an energy profile as shown on the right.
The energy profile on the right is that of an exothermic reaction.
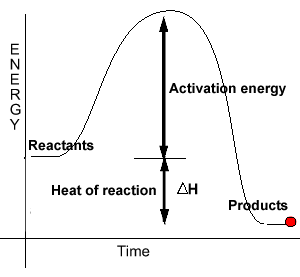
Catalysts introduce alternative reaction pathways with significantly lower activation energies than the uncatalysed reactions. Since the activation energy for this new pathway is less than the activation energy of the normal pathway more molecules are able to take part in fruitful collisions.
Notice how the energies
of the reactants and products remain unaffected by the introduction of
a catalyst. Therefore a catalyst does not change the heat of reaction.Consider the analogy below.
Suppose a mountain divides two towns so that the only way for people to get from one town to the other is over the mountain. Only the most energetic people will be able to get from one town to the other.
Now consider a tunnel cut through the mountain. Many more people will now manage to get from one town to the other by this easier route. The tunnel acts by providing an easier path from one town to the other. One could say that it provides a path wih a lower activation energy than going over the mountain.
The mountain is sitll there it is just that the tunnel has provided an alternative pathway. Some energetic people will still choose to climb the mountain.
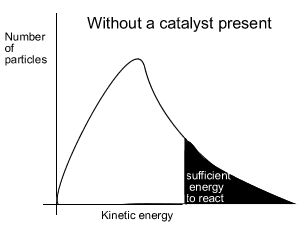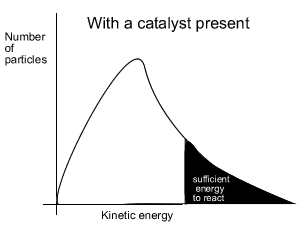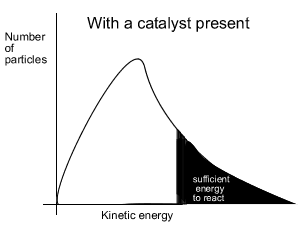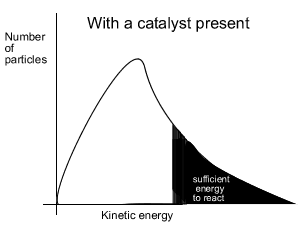
When a catalyst is used, to lower the activation energy, a greater proportion of particles has sufficient energy to now break bonds. With a greater proportion of particles now able to react the reaction proceeds at a faster rate.
A catalyst is not used up during a reaction.
Many inorganic catalysts are used to increase the rate of commercially important reactions. Two such reactions involve the production of ammonia by the Haber process and the production of sulfuric acid.
In the Haber process, an
iron catalyst is used to increase the rate of the exothermic reaction
given below.
3H2(g) + N2(g) => 2NH3(g)
In the absence of a catalyst temperatures around 3,000 oC
are needed to break the bonds within the hydrogen and nitrogen molecules.
During production of sulfuric acid by the Contact process a vanadium oxide is used as a catalyst. The reaction given below proceeds at a fast rate without the need for high pressure and high temperatures, both of which can increase, significantly, the cost of production.
2SO2(g) + O2(g) => 2SO3(g)