Use this formula
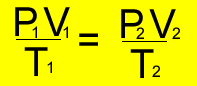
Click to hide the clues
Step 1 Decide what formula you should use. Look at the variables that are given. It is clear that the amount of gas is a constant.
Step 2 Let P2 = 2 P1
Step 3 Solve for V2 Get an expression
in terms of V1
Click to hide the clues
Use this formula
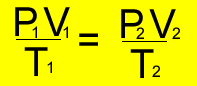
Click to hide the clues
|
Applying the right formula to problem
solving.
|
|
| It is often necessary to select the appropriate formula to solve a given problem. We have covered the formulae listed on the right. It is a matter of selecting the right formula given the information in the problem. Below are four examples with detailed solutions. Attempt all four then click on the solutions to see if you were correct. |  |
|
On an unknown planet a pure
sample of hydrogen gas is placed in 450 litre balloon at a pressure of
80 kPa at a temperature of 23oC and allowed to ascend skywards.
At an altitude of 2,330m the volume of the balloon is recorded to be 350
litres and the pressure 30 kPa. What is the temperature at 2,330m?
|
|
|
The tyres of a fighter jet
have a volume of 45 litres each. Each tyre is pumped up to a pressure
of 410 kPa at 22oC with pure nitrogen. Upon landing the tyres
heat up to 117oC . The tyre manufacturer recommends that the
tyres not be inflated beyond 600 kPa as a blow-out is likely to occur.
What mass of nitrogen is placed
in each tyre? |
|
|
A 7.5 litre sample of oxygen
gas at 284oC was heated, At what temperature would the volume
be 10.0 litres assuming that the pressure remains constant? |
|
| A
gas sample is stored in a cylinder at 284oC. If the pressure
exerted on the gas is doubled and the gas is then heated to 354oC
by what factor does the volume of the gas change? Clues |
|