Food chemistry
Fats and oils
Fat is the general
name given to a large number of organic compounds. Such organic molecules
belong to a larger group of biological compounds known as lipids.
Lipids are formed from carbon, hydrogen and occasionally oxygen. They
are non-polar and therefore not soluble in water.

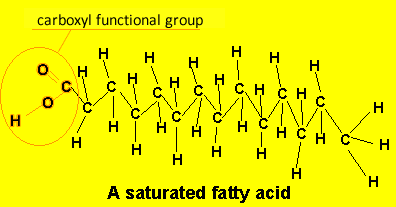
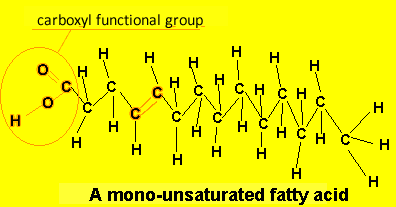
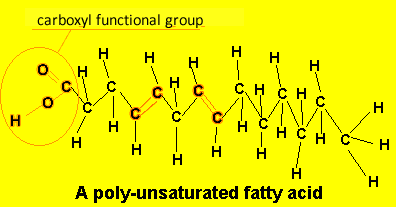
A poly-unsaturated fatty acid has more than one carbon-carbon double bond. eg Linoleic acid
Due to the presence of double bonds, poly-unsaturated fatty acids tend to be more reactive than their saturated cousins. The double carbon-carbon bond also causes the chains to be bent. This prevents close packing of the hydrocarbons and the dispersion forces acting are not able to exert much of an attraction. Therefore, polyunsaturated fatty acids have a lower melting temperature than saturated fatty acids and tend to exist as oils at room temperature.