Enthalpy of solution
of copper (ii) sulfate pentahydrate
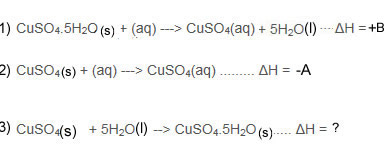
Using Hess's Law you will then work out the ΔH of equation 3.
This has two parts.
Part one reverse equation 1
Part two add it to equation 2.

72.468 grams

Step 2 Measure accurately about 23.0 g of copper sulfate pentahydrate (CuSO4.5H2O )
95.76 - 72.468 = 23.29 grams


Step 4 Measure the temperature of the water.
18.10 oC


Step 6 Record the final temperature of the water.
15.50 oC
Step 7 Calculate the ΔH. of the reaction below
CuSO4 .5H2O (s) + (aq) => CuSO4(aq) + H2O(l) ΔH =
Step 1 calculate the mol of copper sulfate pentahydrate
=> 23.29 / 250.0 = 0.0932
Step 2 Calculate the change in temperature ΔT (18.10 - 15.50 ) = 2.6 oC
Step 3 Calculate the amount of energy released
=> Energy = 4.18J/g/C X 100g X 2.6
oC = 1087 J
Step 4 Calculate the ΔH = 1087 / 0.0932 = + 11.7 kJ/mol