http://web.lemoyne.edu/~giunta/chm151L/stoichiometry.html
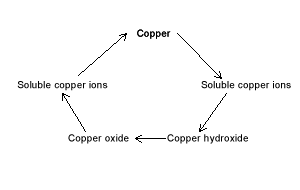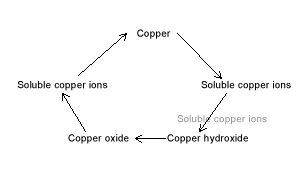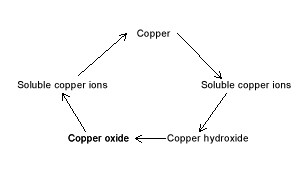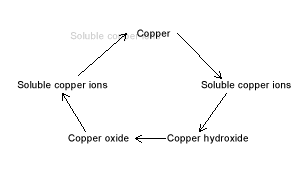
In this set of reactions with copper we will see the formation of copper ions from solid copper metal. Further chemical reactions will transform the copper ions into copper hydroxide(Cu(OH)2) and then into copper oxide (CuO). Copper oxide is transformed back into copper ions and finally back into copper metal. The mass of copper metal we start with should be the same as the mass of metal we finish with.
The entire process:
-
demonstrates the law of "Conservation of mass". That is, atoms can not be created or destroyed during chemical reactions. They are simply rearranged to form new compounds:
-
Illustrates a variety of substances of which an element can be part of;
-
offers experience in laboratory techniques, such as, safety, filtration and quantitative techniques.
Material used:
-1.5 g of copper turnings;
- 5.0 g of zinc;
- 50 mL of 3M sulfuric acid;
- 15 mL of 7M nitric acid;
- 40 mL of 6M sodium hydroxide;
- 250 mL beaker;
- 2 X 50 mL measuring cylinder;
- filter paper, funnel, conical flask, bunsen burner, heat mat, gauze mat, electronic balance, stirring rod, watch glass.
Copper metal => copper ions + nitrogen dioxide + water
Cu (s) + 4HNO3 (aq)--> Cu(NO3)2(aq) + 2NO2 (g) +2H2 O (l)

Copper nitrate + sodium hydroxide => copper hydroxide + sodium nitrate
Cu(NO3)2(aq) + 2NaOH (aq) => 2NaNO3 (aq)+ Cu(OH)2 (s)

Copper hydroxide => copper oxide + water
Cu(OH)2 (s) => CuO(s) + H2O(l)

Copper oxide + sulfuric acid => water and copper sulfate
CuO(s) + H2SO4(aq)=> H2O(l) + CuSO4(aq)

Copper sulfate + zinc metal = copper metal + zinc sulfate
CuSO4(aq) + Zn(s) => Cu(s) + ZnSO4(aq)
