Rust prevention
Sacrificial anodes |
| Iron is a cheap
and strong metal to use as a construction material. It has one basic flaw
however, it is very reactive. It will quickly rust, under the right conditions,
leaving behind a fine brown powder known as iron oxide(rust). We can prevent
iron from rusting by many different methods. The most common way is to paint
the metal and prevent oxygen from reacting with it. This method is very
expensive as coats of paint need to be applied frequently. If the surface
coating of paint is scratched rusting will take place immediately. This
is not the preferred method of rust prevention for giant ships that often
scrape against rocks, sand and barnacles. Instead, a sacrificial
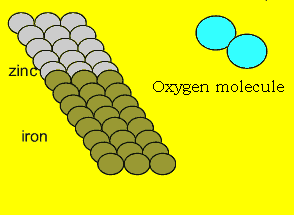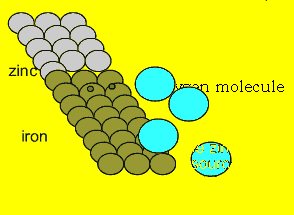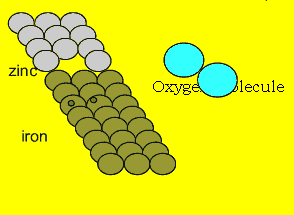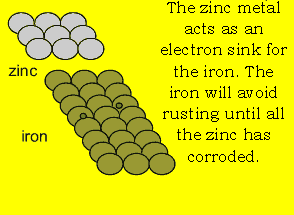
anode is used. A sacrificial anode is a block of metal that
is more reactive than iron. The more reactive a metal is the easier it gives
away electrons. This reactive block of metal acts as a source of electrons
for the iron. When oxygen takes electrons from the iron during the process
of rusting iron atoms simply take electrons from the reactive metal. |
If we wrap zinc
around an iron nail the nail is protected from rusting. Notice that the
iron does not have to be completely covered in order to be fully protected.
This is a real advantage to using a sacrificial anode.
Notice also the white flakes
appearing on the zinc and at the bottom of the test tube. Corrosion is
occurring at a great rate but its not iron but rather zinc that is rusting.
Simply put, the zinc is sacrificing itself to protect the iron hence the
name sacrificial anode.
|
|
|
The animation
on the left shows how electrons flow from zinc to the iron. This process
occurs until all the zinc has corroded or the two metals are no longer in
contact. |
| The hulls of large sea vessels
use small zinc blocks placed at regular intervals to prevent rust. Periodically
these zinc blocks have to be replaced as they corrode away. |
 |
|
Oil rigs use
blocks of aluminium at the foot of every supporting pillar to prevent corrosion
of the iron metal. Aluminium is
very reactive metal. |
Metal
"X" is more reactive than metal "Y".
Both metals are very strong and cheap to produce. A bridge is to be constructed
from these two metals. Discuss which metal you will use for the main structure
and which will form the sacrificial anode. |
|