Synthesis of guncotton
SAFETY -NOTE, THE PREPARATION OF GUNCOTTON MUST BE DONE IN A FULLY FUNCTIONAL FUMEHOOD
You
will need:
- concentrated sulfuric acid
- concentrated nitric acid
- cotton wool
- gloves
- tongs
- goggles
- 200 ml beaker
THIS MUST BE DONE IN A FUMEHOOD
Pour 30 ml of concentrated sulfuric acid in a 200 ml beaker. Cool the sulfuric acid by placing the beaker in ice cold water. Now slowly add 30 ml of nitric acid. Mixing the acids produces a great deal of heat.

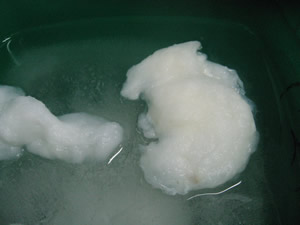
Place a small amount of cotton
wool, as seen in the video below, in the acid mixture until it is completely submerged. Allow the cotton
wool to stand for 15 minutes.
Remove the guncotton and rinse it thoroughly under cold water.
Allow to dry over a 24 hour period.
Place a small piece of guncotton in an evaporating dish and ignite with
a candle attached to a meter ruler.
Click
to see the reaction. This sample of guncotton, shown in the video, was
not completely dry and therefore the reaction was not complete with solid
products left unreacted.
Notice how the reaction produces gaseous products and very little solid
material.
How is the rate of burning different.burning.

Chemical reactions produce products with totally new properties than the reactants.
Consider the video on the right. It shows normal cellulose cotton wool on the left and cellulose cotton wool after it has been bathed in concentrated sulfuric and nitric acids.
Describe the differences?
Safety notes
THE PREPARATION OF GUNCOTTON MUST BE DONE IN A FULLY FUNCTIONAL FUMEHOOD. Always test to see if the fumehood works before commencing the preparation.
Mixing of acids is highly exothermic
(gives off heat).
Guncotton ignites very easily when exposed to a naked flame. Once prepared
do not store all the material must be used. Guncotton can be kept underwater
indefinitely, but this is not recommended.