Chemical snakes worksheet
Uses for alginate range from the food industry to wound dressings, medicines and the dental industry where it is used as an impression material. Calcium alginate is used in dressing for slow healing wounds, such as ulcers which continue to bleed for a long time. When in contact with body fluids, such as blood, calcium alginate exchanges sodium ions for calcium ions. Calcium ions are part of the natural clotting mechanism of the body and therefore assist in clotting of the blood and encourages healing.
Alginate is a common food additive, it can be identified on the label by the symbol E400. It is used as a thickener, stabiliser and gelling agent, found in ice cream and most other foods that require a thickening agent.
Sodium alginate is a polymer composed of carbohydrate molecules linked end to end. It is extracted from brown seaweed and kelps where it is used as one of the structural polymers that build the cell wall of these plants.
When sodium alginate is put into a solution of calcium ions, the calcium ions replace the sodium ions in the polymer as shown on the right.
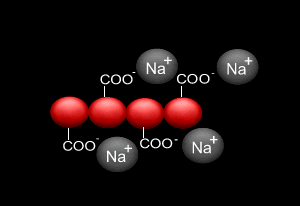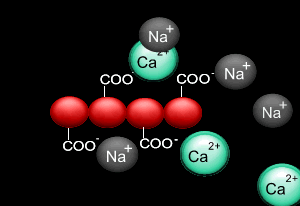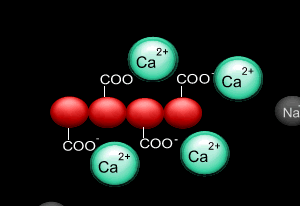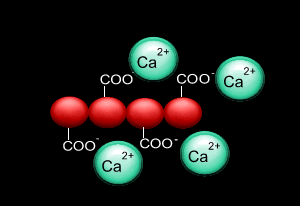
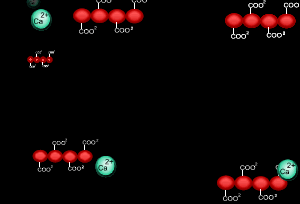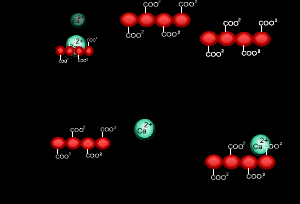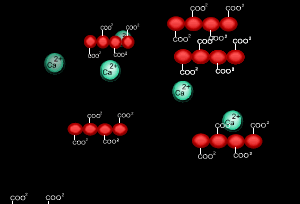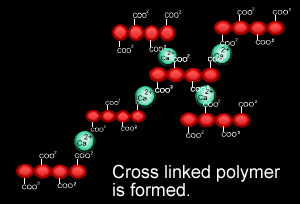
1) Why does the calcium ion replace the sodium ion?
2) Why are cross links formed between the polymers and the calcium ion and the sodium ion?
3) Place some of the snakes in a solution of sodium chloride (salt solution). What do you observe? Can you explain what is happening?
4) Find five foods that contain sodium alginate. Why was sodium alginate added to each food?