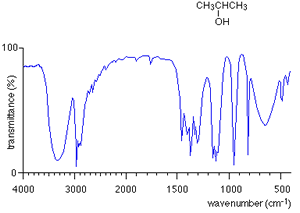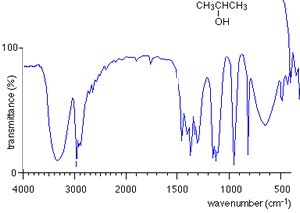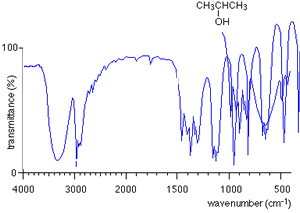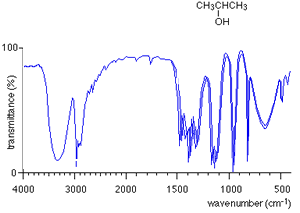
For every compound a very complicated series of absorptions occur between wavenumbers 500 to 1500 due to a variety of bending and stretching within the molecule. This region is called the finger print region. Each compound has a unique set of troughs within the fingerprint region that can be used to identify the molecule. It is hard to identify individual troughs due to specific bonds in the fingerprint region.
Take the IR spectra of propan-1-ol and propan -2-ol as pictured on the right. You can see that both compounds have similar bonds and therefore the troughs around wavenumber 3000 are also similar. However, compare their fingerprint regions between 1500 and 500 and one can see an immediate difference. The fingerprint region can therefore be used to identify the compounds.
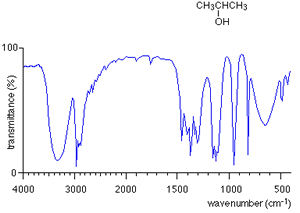
How do we use an IR spectrum? We can identify specific bond types within the molecule by looking for specific troughs at certain wavenumbers outside the fingerprint region. This way we know what type of compound it is by looking for the type of bonds it contains.