The concentration of strong acid in a solution will be equal to the concentration of hydronium ions as the reaction represented by the equation below goes almost to completion.
![]()
The concentration of strong acid in a solution will be equal to the concentration of hydronium ions as the reaction represented by the equation below goes almost to completion.
![]()
![]()
![]()
|
pH
|
||||||||||||||||||||||||
|
pH is a very convenient scale to measure large variations of [H3O+] concentrations. pH is defined
by the expression below |
||||||||||||||||||||||||
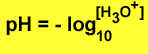 |
||||||||||||||||||||||||
| The above expression can be rearranged to give: | ||||||||||||||||||||||||
| Example1. Calculate the pH of a 0.1M HCl solution. Step 1 find the concentration of hydronium ions. Since HCl is a strong acid the reaction represented by the equation below has a very high Ka value therefore the reaction proceeds almost to completion. |
||||||||||||||||||||||||
| The concentration of HCl should be equal to the concentration of hydronium
ions. Step 2 |
||||||||||||||||||||||||
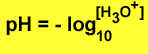 |
||||||||||||||||||||||||
| Example 2. Calculate
the concentration of hydronium ions in a sample of a lemon soda drink of pH 3.0. Step 1 Apply the formula below |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||