Separating oil from food
(senior chemistry investigation).
The video on the right shows oil that came from a given mass of a popular brand of blue berry muffin. Oil was isolated following the procedure given below.






The evaporating dish is then wash and thoroughly dried.
The filtrate is then poured into the evaporating dish and allowed to evaporate overnight in a well ventilated fume cupboard.


1. Calculate the percent, by mass, of oil in the muffin.
i. Construct a table of data.
ii.
Show all calculations
2. Consider the image on the right. It is a pre-packaged muffin sold at a convenience store similar to the one tested. Calculate the mass, in grams, of oil present in one whole muffin, as shown on the right.
3. Using some or all the terms listed below, explain some of the observed physical properties of the food dye.
- hydrophobic.
- dipole-dipole.
- polar
- non-polar
- intermolecular
4. The procedure shown above attempted to isolate the oil from a given mass of muffin.
i
Suggest two possible errors with the procedure given above and explain the impact it would have on the final result of the percentage, by mass, of oil content.
ii.
Offer two ways to improve the procedure.

5. Consider the label on the right.
a. Identify 3 risks associated with the use of acetone.
b. For each safety risk, suggest one way to mitigate the risk.

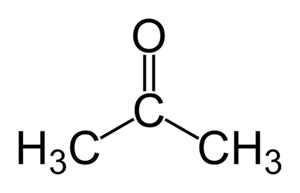
6. With reference to the chemical structure of acetone explain how acetone is a solvent for both hydrophobic and hydrophilic substances.
7. With reference to the safety data given in question 5. above acetone is a volatile substance that must be handled in a well ventilated laboratory.
i.
Compare the flash points of propane and acetone. Explain your reasoning.
ii. Compare the solubilities in water of acetone and propane. Explain your reasoning.
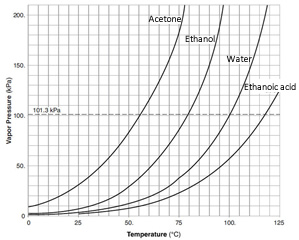
8. Vapour pressure is the pressure exerted by molecules leaving the liquid and entering the gaseous state. Vapour pressure depends only on the temperature of the liquid and not on the volume of the container or the amount of substance.
i.
Given the following liquids acetone, ethanol, ethanoic acid (vinegar) and water explain the trend in vapour pressure shown by the graph on the right.
ii. A pure 5.00 g sample of acetone (58.0 g/mol) is placed in a sealed 1.00 litre vessel at 75 oC. Will there be liquid acetone left in the container after 2 hours? Justify your answer with a calculation.
9. Consider the image of the lipid isolated from the muffin. Is a saturated or unsaturated fatty acid used in the making of these muffins? Explain.
10. The table on the right indicates the results achieved by four groups conducting the investigation on different days, following the same procedure but using different, scales.
Answer the following questions and justify your answer.
i. Are the results from group1 precise?
ii. Are the results valid?
iii. Are the results obtained reliable?
| Group | trial 1 % (m/m) |
trial 2 % (m/m) |
trial 3 % (m/m) |
Average % (m/m) |
| 1 | 9.8 | 9.9 | 9.7 | 9.8 |
| 2 | 12.3 | 12.4 | 13.7 | 12.8 |
| 3 | 7.8 | 9.4 | 10.1 | 9.1 |
| 4 | 15.3 | 7.9 | 10.3 | 11.2 |